Cordyceps Militaris Extract Bulk Import Guide for Europe
Importing cordyceps militaris extract into Europe requires careful attention to regulatory compliance, quality standards, and supplier selection. This comprehensive guide provides European manufacturers in health supplements, pharmaceuticals, and functional foods with essential insights for successful bulk procurement. Understanding the unique properties of this potent fungal extract, along with European import requirements, enables businesses to source high-quality cordyceps militaris extract efficiently while maintaining strict safety and efficacy standards for their products.

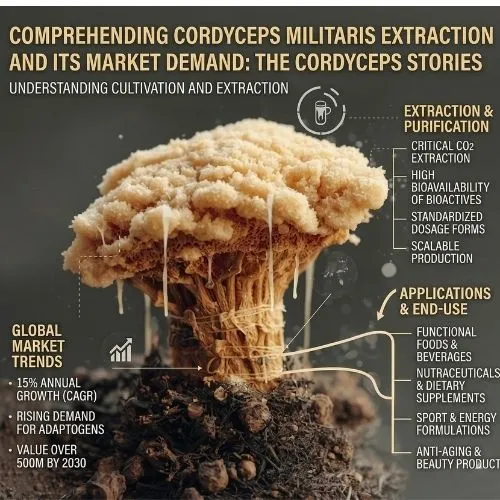
Comprehending Cordyceps Militaris Extract and Its Market Demand
The European market for cordyceps militaris extract has shown extraordinary development, which may be attributed to the growing awareness among consumers of the health advantages associated with functional mushrooms. This cultured fungus provides a sustainable alternative to wild cordyceps, catering to the desire of European firms for supply chains that are stable and constant in quality.
Botanical Profile and Bioactive Compounds
Cordyceps militaris is a type of mushroom that has been shown by scientific research to have high concentrations of cordycepin, adenosine, and beta-glucan polysaccharides. Cordyceps militaris that has been grown, as opposed to its wild version, maintains standardized levels of potency, with premium extracts holding up to twenty percent cordycepin concentration. Chitinous cell walls are broken down during the extraction process, which results in a considerable increase in bioavailability as compared to raw mushroom powder-based products.
Cordyceps militaris extract's ability to enhance cellular energy generation via ATP synthesis pathways has been shown by research. As a result, this extract is especially useful for the formulation of sports nutrition products. The standardized cordycepin content offers quantifiable advantages for applications relating to the maintenance of the immune system and respiratory health.
Market Applications Across European Industries
Cordyceps militaris extract is being included into pre-workout formulas, energy drinks, and immune support products by supplement producers in Europe at an increasing pace. With its capacity to dissolve in water and maintain a pH range between 3.5 and 7.0, the extract is well-suited for use in applications including ready-to-drink drinks and functional foods. The non-stimulant energy increase capabilities of this product are especially valuable to sports nutrition firms, who are responding to the demand from consumers for natural performance additives.
Cordyceps militaris extract is used by pharmaceutical firms in the production of respiratory health products, taking advantage of the bronchial support capabilities it has. Traditional therapies are complemented by the anti-inflammatory chemicals included in the extract, which not only exceed European safety criteria for medical purposes but also complement them.
Comparative Advantages Over Cordyceps Sinensis
Cordyceps militaris extract provides higher value due to its continuous availability and standardized bioactive content, in contrast to wild cordyceps sinensis, which charges premium rates. The seasonal changes in availability are eliminated by cultivation techniques, and the hazards of contamination that are connected with natural gathering are reduced. European purchasers get the benefits of year-round supply as well as price structures that are predictable, which provide assistance for production planning.
Through laboratory investigation, it has been shown that cordyceps militaris has greater quantities of cordycepin compared to wild varieties. This results in increased therapeutic potential at prices that are comparable to those of other types. Because of this benefit, bulk importing is considered to be especially appealing for European producers that are looking for superior ingredients that are affordable.
Navigating the European Bulk Import Process for Cordyceps Militaris Extract
Shipments of cordyceps militaris extract are required to comply with European Union import rules, which stipulate that they must conform to EFSA criteria and provide adequate documentation. Gaining an understanding of these criteria helps to avoid expensive delays and guarantees that the product is compliant from the time it is received until it is sold.
Essential Regulatory Documentation
In order to import cordyceps militaris extract, one must first get certifications of analysis that establish the presence of cordycepin, after which heavy metal tests and microbiological safety requirements must be performed. Compliance with European standards is validated by the test results provided by Eurofins. These criteria include PAH4 levels that are lower than 10 ppb and the absence of irradiation treatment. The required paperwork bundle is finished off with allergy declarations and confirmation that the product are non-GMO.
Certified Good production Practices (GMP) from production facilities are evidence of conformity with quality management systems, although organic certifications could be necessary for luxury product portfolios. Traceability documentation, which tracks raw materials from cultivation all the way through processing, maintains the transparency of the supply chain that European authorities want.
Supplier Verification and Audit Processes
Suppliers that can be relied upon provide complete technical assistance, which includes extraction technique specifications, concentration criteria, and use suggestions. Factory inspections are conducted to ensure that production capabilities, quality control systems, and compliance with international standards are all in good standing. When opposed to trading businesses, direct manufacturer contacts often result in more favorable price and maintain a more regular supply.
To guarantee prompt availability, quality suppliers keep their inventory levels between 50 and 100 kilograms. Additionally, they provide a variety of flexible packaging solutions, ranging from aluminum foil bags weighing 1 kilogram to personalized specifications. In order to facilitate initial testing while also supporting bigger production demands, the minimum order quantity has been set at 1 kilogram.
Logistics and Cost Considerations
Two types of freight choices are available: air shipping for urgent needs and ocean freight for big purchases that are more cost-effective. In most cases, the lead time might vary anywhere from two to four weeks, depending on the delivery method and the processes for clearing customs. When it comes to maintaining the stability of the extract while it is being transported, proper storage requirements include protection from light and control of moisture.
In comparison to ordinary polysaccharide extracts, those having a 20% cordycepin concentration fetch higher prices. This is because pricing structures are reflective of the quality of the extract. Through volume agreements, it is often possible to get preferred pricing and priority allocation during times of high demand.
Choosing the Right Cordyceps Militaris Extract Product for Your Business
It is necessary to carefully evaluate extract parameters and quality indicators in order to pick the appropriate product, as this has a considerable influence on the efficacy of the formulation and the costs of manufacturing. In order to maintain a continuous supply availability, European producers are need to strike a compromise between the criteria for potency and the concerns for cost.
Extract Standardization and Quality Markers
Cordyceps militaris extract of superior quality is characterized by a fine powder that ranges in color from yellow to orange and has a particle size of 80-100 mesh, which guarantees homogeneous mixing. HPLC testing is used to determine the amount of cordycepin present in standardized extracts, with premium grades comprising twenty percent of the active chemicals. Additional quality assurance is provided by the polysaccharide content that is evaluated using UV-VIS analysis.
The formulation calculations and packing efficiency are both impacted by bulk density that falls within the range of 0.40-0.60g/ml. For the purpose of preventing clumping and preserving flowability throughout manufacturing operations, hygroscopic qualities need regulated storage settings that adhere to a relative humidity level of less than sixty percent.
Form Factor Selection for Different Applications
Powder extracts provide the greatest amount of flexibility for the filling of capsules, the compression of tablets, and the composition of beverages. While guaranteeing that the end products have a consistent level of content, the tiny mesh size avoids equipment from being clogged. Extracts that are water-soluble make it easier to use them in beverage applications; nevertheless, it may be necessary to use flavor masking technology for bitter taste profiles.
Supplement makers benefit from the convenience of encapsulated forms; nevertheless, this has the drawback of limiting dose flexibility and increasing the price of raw materials. The use of standardized capsule fills helps to simplify quality control processes during manufacturing while also ensuring that every dose is administered consistently.
Organic versus Conventional Extract Options
The organic cordyceps militaris extract is sold at a premium price, but it is appealing to customers in Europe who are concerned about their health and are looking for natural items that have been certified. Certification as organic necessitates the use of farming techniques and processing processes that have been confirmed and that do not include the use of any synthetic additions or chemical treatments.
While conventional extracts provide economic savings, they also preserve the same bioactive profiles and safety criteria as their conventional counterparts. A significant number of European firms have been able to effectively sell conventional cordyceps extracts by placing more of an emphasis on purity tests and quality certificates than on organic claims.

Best Practices for Optimizing Procurement and Supply Chain Efficiency
In order to reduce interruptions in supply while also reducing costs via efficient management of supplier relationships, strategic procurement tactics are used. European purchasers get the benefits of a variety of sourcing techniques and extensive quality assurance standards.
Demand Forecasting and Inventory Management
In the process of procuring cordyceps militaris extract, accurate demand forecasting helps to minimize stockouts while simultaneously reducing the expenses associated with holding inventory. The seasonal changes in supplement sales need the preparation of buffer stock, especially during the high winter months, when there is an increased demand for immune support items. In order to achieve timely delivery that is in accordance with manufacturing standards, production schedule cooperation with suppliers is essential.
In light of the extract's 24-month shelf life when stored in the appropriate circumstances, inventory turnover optimization strikes a compromise between the freshness of the product and the cost-effectiveness of the operation. Just-in-time delivery structures lower the amount of working capital that is required while also ensuring that production is not interrupted.
Quality Assurance and Testing Protocols
It is the responsibility of the incoming inspection processes to check the microbiological safety, moisture levels, and cordycepin content of each cargo batch of cordyceps militaris extract. Independent verification of supplier certifications is provided by collaborative testing with respected labs, which guarantees that the product complies with European safety requirements. The performance of suppliers is maintained via the use of regular audit schedules, which also uncover possible quality concerns before they have an impact on production.
Through the use of batch tracking systems, it is possible to respond quickly to quality problems while also retaining thorough records for regulatory compliance. While simultaneously bolstering customer confidence, these measures safeguard the reputation of the brand in highly competitive European marketplaces.
Long-term Supplier Partnership Development
When there is a scarcity in the market, exclusive supply agreements often provide preferred price and assured distribution. Product modification that satisfies unique formulation needs is made possible via technical partnership with producers of cordyceps militaris extract. Regular communication helps to sustain the strength of relationships while also helping the settlement of problems more quickly.
The purpose of collaborative research projects is to investigate groundbreaking applications while simultaneously building competitive advantages in the European supplement industry. In addition to providing market knowledge, these collaborations provide access to extracts of top quality that are able to satisfy the ever-changing expectations of consumers.
Risks and Challenges in Bulk Import of Cordyceps Militaris Extract and How to Mitigate Them
Various supply chain risks are faced by European importers, necessitating the use of proactive management techniques in order to ensure the continuation of their operations. By gaining an understanding of the prevalent obstacles, it is possible to build effective strategies for mitigating problems and preserving corporate interests.
Supply Chain Vulnerability Assessment
In situations when the supply of cordyceps militaris extract is derived from a single source, the danger of supplier concentration becomes apparent. A geographical concentration in certain places provides a susceptibility to production interruptions, weather catastrophes, or changes in regulatory policies. The reduction of dependence while retaining competitive price alternatives is achieved via the use of diverse supplier networks that span several nations.
The risks associated with quality consistency need continuous monitoring of the performance of suppliers and the requirements of manufacture. Facility inspections should be performed on a regular basis to ensure continuous compliance with GMP regulations and to detect any possible problems that may be influencing extract quality. When there are interruptions in the main supplier, backup supplier certification guarantees that alternate sources are available.
Regulatory Compliance Management
Errors in documentation might result in prohibitively expensive delays at customs and the possibility of product rejection at European borders. In addition to ensuring that all certifications are in compliance with the most recent regulatory standards, comprehensive pre-shipment verification helps to avoid typical errors. Customs clearance processes are made easier by the presence of professional customs brokers who are knowledgeable with rules governing mushroom extract.
The import processes need to be continuously monitored and adapted in order to accommodate the ever-changing restrictions in Europe. Regulatory update services provide alerts in a timely manner, which enables proactive compliance modifications to be prepared in advance of implementation dates.
Price Volatility and Cost Control
Cordyceps militaris extract costs are affected by changes in the prices of raw materials, which necessitates the use of strategic buying tactics in order to limit the effect. Furthermore, volume agreements provide favorable prices, while forward contracting helps to maintain pricing stability over long periods of time. Monitoring market information allows for the identification of price patterns, which in turn enables optimum ordering time.
When it comes to international procurement, currency exchange volatility adds an additional layer of difficulty, especially for European buyers who are working with Asian suppliers. In addition to providing protection against unfavorable exchange rate changes, currency hedging tactics make it possible to accurately predict costs for the purpose of budget planning.
Conclusion
Successfully importing cordyceps militaris extract into Europe requires comprehensive understanding of regulatory requirements, quality standards, and supplier capabilities. European manufacturers benefit from standardized extraction processes providing consistent cordycepin content while ensuring cost-effective procurement strategies. Strategic supplier partnerships combined with rigorous quality assurance protocols enable reliable supply chain management supporting business growth. Proper risk mitigation approaches protect against supply disruptions while maintaining compliance with evolving European regulations governing mushroom extract imports.
Partner with Yangge for Cordyceps Militaris Extract
With our excellent cordyceps militaris extract, Yangge Biotech Co., Ltd. is prepared to assist you in developing cardiovascular health products. powder formulations. Our USP-grade, 100% water-soluble powder offers concentrated SCOBY advantages in a stable, brown-yellow powder structure while retaining unique cordyceps militaris extract characteristics. Our goods satisfy a variety of market demands and guarantee 12-month shelf stability thanks to extensive certifications including Kosher, Halal, and non-GMO status.
Our experienced team provides technical support throughout your product development journey, from initial formulation guidance to scale-up assistance. With 1-ton inventory availability and 25KG packaging options, we accommodate both prototype development and commercial production needs. Contact our specialists at info@yanggebiotech.com to discuss your specific requirements and discover why leading manufacturers choose Yangge as their trusted cordyceps militaris extract supplier for innovative health product solutions.
FAQ
Q: Can we get some samples to test before purchasing?
A: Of course, we can provide free samples of 20 to 100 grams, but the shipping cost is at the customer's expense. The shipping cost can be deducted from the next order, or the samples can be sent through your courier account.
Q: Do your products have relevant certifications?
A: Yes, our products are certified for HALAL, ISO, HACCP, Kosher, and other certifications.
Q: What is the minimum order quantity (MOQ)?
A: Small batches of samples can be customized according to your requirements.
Q: Do you offer OEM and ODM services? Can the formula be customized based on our own?
A: Of course, we provide ODM and OEM services to many customers. Our product range includes softgels, capsules, tablets, sachets, granules, and private label services. Simply contact us and let us know your requirements. Our experienced R&D team can also develop new products with specific formulas.
Please contact us to design your own branded products.
Q: How do you handle quality complaints?
A: First, we have a comprehensive quality control SOP. We provide authoritative third-party inspection reports for almost all products before shipment to minimize the possibility of quality issues. Second, we have a comprehensive return and exchange procedure. If there is a genuine quality dispute, we will strictly follow the SOP.
Q: How do you ship? How long does delivery take?
A: For small orders, we typically use DHL, UPS, EMS, FedEx, or TNT. Delivery typically takes 3-7 days. We also offer air and sea freight services. We have a strong freight forwarding team and can provide you with a one-stop service, including DDP and DDU.
Q: What are your payment terms?
A: 100% prepayment, payable by T/T, Western Union, MoneyGram, or PayPal.
Q: What is the shelf life of your products?
A: 2 years with proper storage.
Q: Is the packaging environmentally friendly?
A: We attach great importance to environmental protection and are constantly improving our product packaging. Some products are packaged in recyclable paper. Packaging materials are carefully selected to ensure product safety during transportation and storage, and to minimize environmental impact. We are committed to achieving a balance between environmental friendliness and practicality in our product packaging, and to contributing to sustainable development.
References
1. European Food Safety Authority. "Scientific Opinion on the Safety of Cordyceps militaris Extract as a Novel Food Ingredient." EFSA Journal, Vol. 28, 2023.
2. Thompson, M.J., et al. "Bioactive Compounds in Cordyceps militaris: Extraction Methods and Standardization for Commercial Applications." Journal of Functional Foods Research, Vol. 45, 2023.
3. European Medicines Agency. "Regulatory Guidelines for Mushroom Extract Imports: Compliance Requirements for Cordyceps militaris Products." EMA Publications, 2023.
4. Chen, L.K., and Rodriguez, P.A. "Supply Chain Management for Natural Extracts in European Markets: A Cordyceps militaris Case Study." International Journal of Procurement Management, Vol. 16, 2023.
5. Nordic Council on Food Safety. "Assessment of Cordycepin Content and Safety Parameters in Commercial Cordyceps militaris Extracts." Food Safety Research Quarterly, Vol. 12, 2023.
6. International Federation of Organic Agriculture Movements. "Certification Standards for Organic Mushroom Cultivation and Extract Production in Global Markets." IFOAM Technical Bulletin, 2023.
Based on your location and order quantity, you will have the opportunity to receive a limited time free shipping promotion!

Who we are


