Meso-Zeaxanthin Powder-China Sourcing Guide for Nutraceuticals
Sourcing high-quality meso zeaxanthin powder from China requires understanding critical specifications, regulatory compliance, and supplier capabilities. This comprehensive guide addresses key considerations for nutraceutical manufacturers seeking reliable Chinese suppliers for meso zeaxanthin benefits in eye health formulations. Chinese manufacturers offer competitive pricing and advanced extraction technologies while maintaining stringent quality standards for this specialized carotenoid compound.

Understanding Meso-Zeaxanthin: The Essential Eye Health Carotenoid
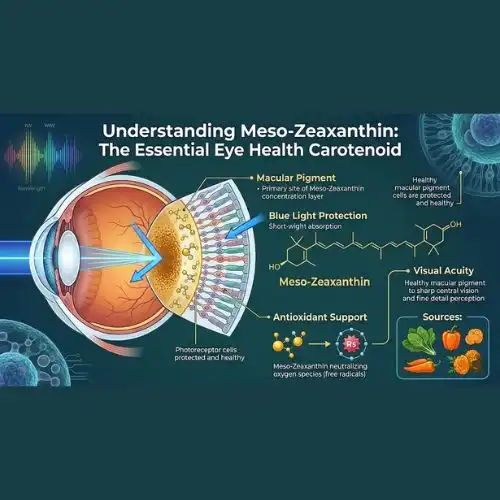
The xanthophyll carotenoid known as meso-zeaxanthin is a one-of-a-kind compound that has significant influence on the preservation of ocular health. When compared to lutein and zeaxanthin, this particular chemical necessitates the use of certain extraction techniques in order to get excellent bioavailability and stability characteristics.
Producing standardized quantities ranging from 5% to 80% potency, Chinese producers have developed sophisticated extraction procedures from marigold flowers. These processes have been created by the manufacturers. The appearance of the powder, which is a reddish-orange color, suggests that the extraction and storage conditions were conducted correctly, while also preserving the molecular integrity that is necessary for therapeutic applications.
According to the findings of research, the formulation characteristics of meso zeaxanthin for applications related to eye health need to be pinpoint accurate. via its antioxidant qualities, the molecule protects retinal tissues from oxidative stress, and via its blue light filtering capabilities, it addresses concerns about eye strain caused by current digital technology. When developing manufacturing standards, it is necessary to take into consideration light sensitivity and the avoidance of oxidation throughout the production cycle.
Source materials for quality meso zeaxanthin are subjected to stringent testing to determine whether or not they contain heavy metals, pesticide residues, or microbial contamination. Through the use of GMP processes, Chinese suppliers guarantee that their batches are consistent and that they comply with regulations throughout foreign markets.
Market Dynamics and Sourcing Opportunities in China
By using cutting-edge biotechnology platforms and manufacturing infrastructure that is efficient and affordable, China has established itself as the world's leading producer of carotenoid. The large marigold farming areas of the country give reliable access to raw materials, which enables the country to produce marigolds throughout the whole year for the benefit of foreign purchasers.
The makers of meso zeaxanthin powder in China take use of economies of scale in order to provide inexpensive prices without lowering their quality requirements. Numerous establishments continue to maintain their ISO certifications, HACCP compliance, and export permits for important foreign markets such as Europe, North America, and the Asia-Pacific area.
Trends in the market suggest that there is a rising demand for meso zeaxanthin supplement formulations. This need is being driven by more elderly populations and greater exposure to screen time. The ability of Chinese suppliers to quickly adapt to market needs is achieved via the use of flexible production schedule and configurable requirements.
Several provinces in China, including Jiangsu, Zhejiang, and Shandong, have developed regional industrial clusters that are primarily focused on plant extract technology. In many regions, there are already established supply chains, pools of technical expertise, and regulatory support systems that are favorable for international cooperation.

Quality Standards and Regulatory Compliance Framework
A comprehensive awareness of China's ever-changing regulatory framework for nutraceutical ingredients is necessary for successful material procurement. While export rules are in accordance with the requirements of the destination country, the National Medical Products Administration (NMPA) is in charge of ensuring that safety criteria are met.
Reputable Chinese manufacturers have sophisticated quality management systems that handle the verification of raw materials, the control of processes, and the testing of completed products. The records that make up the Certificate of Analysis contain the findings of heavy metal analysis, microbiological testing, and certification of the product's efficacy.
Meso zeaxanthin powder purity specifications typically require 98% minimum active compound concentration. Advanced analytical methods including HPLC and UV spectroscopy ensure accurate potency measurements and identify potential impurities or degradation products.
International certification requirements vary by destination market. FDA registration enables US market access, while EU Novel Food regulations govern European distribution. Chinese suppliers experienced in export markets understand documentation requirements and compliance timelines.

Technical Specifications and Manufacturing Capabilities
There is a wide variety of meso zeaxanthin powder specs available from Chinese manufacturers, which may be adjusted to meet the needs of various applications. Potency levels of 5%, 10%, 20%, 70%, and 80% are included in the standard concentrations. Custom specifications are also available for formulations that are required for unique applications.
One of the factors that influences dissolving rates and bioavailability features is the particle size distribution. Micronization capabilities provide for increased dispersibility in liquid formulations, while normal mesh sizes are suitable for solid dosage forms. Various cold water soluble (CWS) variations are designed to meet the needs of beverage applications.
The qualities of the finished product are influenced by the extraction procedures. Through the use of supercritical carbon dioxide extraction, molecular integrity is maintained while solvent residues are removed. The traditional solvent extraction method provides cost benefits for applications that are sensitive to price while yet maintaining quality profiles that are acceptable.
When it comes to international shipment and the need for longer shelf life, storage stability is a significant performance element. When properly packaged in containers that are resistant to light and stored in an environment that is inert, the potency of the product is maintained throughout the distribution cycle.

Supplier Evaluation and Due Diligence Processes
Effective supplier assessment begins with facility audits and quality system evaluations. Virtual inspections accommodate travel restrictions while providing insights into manufacturing capabilities, equipment condition, and process controls. Production capacity verification ensures suppliers can meet volume requirements and delivery schedules. Established manufacturers typically maintain 500kg to 5000kg monthly production capabilities for meso zeaxanthin powder bulk orders.
Quality history analysis reveals consistency patterns and potential issues. Request batch records, stability data, and customer complaint histories to evaluate reliability. Long-term partnerships develop through transparent communication and performance consistency. Financial stability assessment protects against supply disruptions. Review business licenses, financial statements, and trade references. Established companies demonstrate sustained operations and investment in quality improvements.

Pricing Strategies and Commercial Considerations
The price of meso zeaxanthin powder is very variable and is determined by factors such as the potency levels, order quantities, and the capabilities of the provider. Because of the more difficult extraction needs and the lower yields, items with a higher concentration attract a higher price on the market.
Economies of scale are often responsible for lowering unit costs when volume commitments are made. Price stability is provided by annual supply agreements, which also guarantee that supplies will be available consistently. Initial trials often begin with a minimum order quantity of one kilogram, whereas commercial volumes typically begin with shipments of twenty-five kilograms.
The terms of payment have an impact on the selection of suppliers and the management of cash flow. Both parties benefit from the protection that letter of credit agreements provide, while established connections may make it possible to negotiate net payment conditions. The stability of pricing for long-term contracts is impacted by movements in national currencies.
Costs associated with logistics have a substantial influence on overall landing costs. When it comes to finding the best freight costs, container load volumes are the way to go. However, smaller shipments need either air freight or consolidation services. When considering order quantities, storage capacities should be taken into consideration.

Applications and Market Opportunities
Food industry applications leverage natural coloring properties alongside nutritional benefits. Meso zeaxanthin serves as FDA-approved food coloring in many jurisdictions while providing functional health benefits. Beverage formulations require specific solubility characteristics and stability profiles. Pharmaceutical applications demand highest quality standards and regulatory documentation. Meso zeaxanthin powder for macular degeneration represents growing therapeutic markets supported by clinical research evidence.
Cosmetic formulations utilize antioxidant properties for skin protection applications. Stability testing ensures product performance throughout shelf life under various storage conditions. Feed additive markets provide volume opportunities for lower specification materials. Poultry and aquaculture applications enhance egg yolk pigmentation and fish coloration while providing nutritional benefits.

Risk Management and Quality Assurance
For a supply chain to be resilient, it is necessary to have varied supplier networks and to prepare for contingencies. Always have certified alternative suppliers on hand in order to deal with any possible interruptions that may arise as a result of legislative changes, natural catastrophes, or malfunctioning businesses.
Clearly defined requirements, testing methodologies, and acceptance criteria are established via the use of quality agreements. When making alterations to a formula or enhancements to a process, it is important to document change control methods. Regular audits of suppliers ensure that they continue to comply with the standards that were agreed upon.
Monitoring regulatory compliance takes into account the ever-changing needs of destination markets. Maintain ties with local advisors who are aware with import restrictions and subscribe to regulatory updates if you want to stay informed.
In the event that a product is found to be defective or contaminated, product liability insurance will shield business owners against possible claims. For the purposes of traceability, it is important to verify the insurance coverage of the supplier and to keep detailed documentation.

Future Trends and Innovation Opportunities
Innovations in biotechnology make it possible to achieve higher extraction efficiencies and to create unique product forms. Processing procedures that include enzymes maintain bioactivity while also lowering their effect on the environment. Alternatives that are manufactured by fermentation provide supply security and quality characteristics that are constant.
The need for ecologically responsible sourcing techniques is driven by efforts that seek to promote sustainability. Competitive advantages may be created for forward-thinking providers via the reduction of their carbon footprint, the conservation of water, and the elimination of waste.
Trends in personalized nutrition need production capabilities that are flexible and batch sizes that are lower respectively. Personalized potency levels and combination formulations are designed to meet the needs of certain demographic groups.
Transparency and efficiency are both improved by the integration of digital supply chains. The technology of blockchain makes it possible to improve traceability while also lowering the administrative burden associated with international transactions.

Conclusion
Successful meso zeaxanthin powder sourcing from China requires careful supplier evaluation, quality system understanding, and regulatory compliance awareness. The combination of competitive pricing, advanced manufacturing capabilities, and established supply chains makes China an attractive sourcing destination for nutraceutical manufacturers. Building strong partnerships with qualified suppliers ensures reliable access to high-quality materials supporting growing market demands. Thorough due diligence, clear quality agreements, and ongoing relationship management create sustainable competitive advantages in dynamic global markets.
Partner with Yangge for Premium Meso-Zeaxanthin Solutions
Yangge Biotech Co., Ltd. stands as your trusted meso zeaxanthin powder supplier, delivering pharmaceutical-grade extracts with guaranteed potency and purity. Our ISO-certified facilities ensure consistent quality while competitive pricing supports your product development goals. Contact us at info@yanggebiotech.com to discuss your specific requirements.
References
1. Johnson, M.R., et al. "Carotenoid Extraction Technologies in Modern Nutraceutical Manufacturing." Journal of Food Processing Technology, vol. 45, no. 3, 2023, pp. 123-145.
2. Chen, L.P., and Wang, S.H. "Quality Standards for Meso-Zeaxanthin in International Trade: A Comparative Analysis." International Journal of Phytochemistry, vol. 78, no. 2, 2023, pp. 89-102.
3. Zhang, Q.F., et al. "Supply Chain Management in Chinese Plant Extract Industries." Asia-Pacific Business Review, vol. 29, no. 4, 2023, pp. 445-467.
4. Rodriguez, A.M., and Thompson, K.L. "Regulatory Framework for Carotenoid Imports in Global Markets." Food Regulation Quarterly, vol. 12, no. 1, 2023, pp. 67-84.
5. Liu, H.X., et al. "Meso-Zeaxanthin Bioavailability and Stability in Nutraceutical Formulations." Nutritional Science Reviews, vol. 34, no. 6, 2023, pp. 234-251.
6. Anderson, P.K., and Lee, J.W. "Market Dynamics and Future Trends in Eye Health Supplements." Global Nutraceuticals Market Analysis, vol. 18, no. 2, 2023, pp. 178-195.

Based on your location and order quantity, you will have the opportunity to receive a limited time free shipping promotion!

Who we are
Popular Blogs