Is kombucha good for people with high blood pressure?
Because of its probiotic composition and bioactive substances that may promote cardiovascular health, kombucha has remarkable promise for those with high blood pressure. As a concentrated version of this fermented tea, kombucha powder provides suppliers and manufacturers with a stable component option that maintains advantageous qualities while offering improved shelf life and formulation flexibility for health-focused product creation aimed at hypertensive customers.

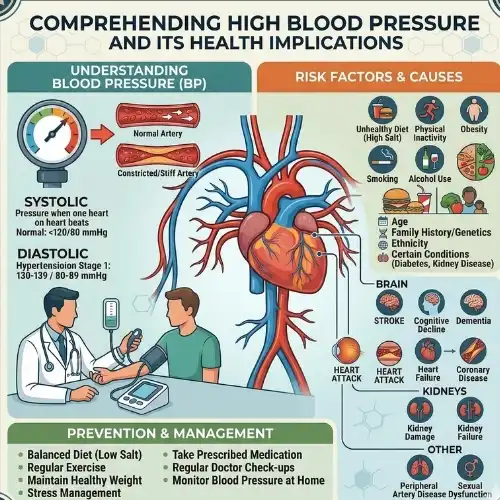
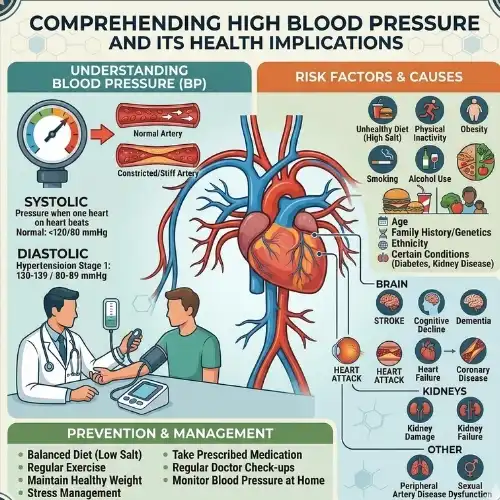
Comprehending High Blood Pressure and Its Health Implications
The medical community refers to high blood pressure, commonly known as hypertension, as a condition that affects more than forty-five percent of the population in the United States. The American Heart Association is the source of this information, and it is said that the number in question is roughly 45 percent. The majority of people are affected by this significant health issue, which is a condition that affects a significant number of people. Hypertension is a condition that is present when the measurements of blood pressure consistently exceed 130/80 mmHg. This condition is an indication of hypertension. The distinguishing aspect of this illness is that it is characterized by high blood pressure. The artery walls are thus subjected to a greater degree of strain as a consequence of this illness, which eventually leads to a significant increase in the likelihood of developing cardiovascular disease.
Clinical Parameters and Risk Factors
To get to the heart of the matter, the pathophysiology of hypertension is primarily composed of a great number of different systems that are related to one another. Instances of these processes include the stiffness of the artery walls, the rise in peripheral resistance, and the dysregulated activity in the renin-angiotensin-aldosterone pathway. The procedures that are being described here are all examples of the same thing. The development of this condition is influenced by a number of different factors, each of which plays a possible role.
Among these causes, the most significant ones include a genetic susceptibility, an excessive quantity of salt in the diet, sedentary lifestyle habits, chronic stress exposure, and changes in the cardiovascular system that occur with advancing age. Because of these components, the clinical image that is produced is a difficult one that requires the use of a large number of various treatment methods. This is because the clinical picture is formed as a consequence of these components.
Commercial Market Implications for B2B Procurement
The worldwide market for hypertension management is exhibiting a strong growth trajectory, which is being driven by the growing understanding among consumers about preventative healthcare measures. This is a positive development for the industry. Currently, the United States of America is working to raise awareness of this issue. Business-to-business procurement specialists are of the opinion that this trend creates a substantial potential for the development of wellness goods that are supported by scientific facts and are beneficial to the cardiovascular system.
Studies that have been carried out on the market reveal that an increasing number of persons are asking for natural alternatives and functional meals that should be utilized in combination with established medical treatments. As a direct result of this, there has been a significant rise in the number of people looking for novel dietary options.
It is necessary to take a cautious approach in order to effectively traverse the regulatory framework that is associated with claims about cardiovascular health. The Food and Drug Administration (FDA) has set several guidelines, one of which is that there must be a clear proof of the health advantages, and another is that the required labeling processes must be followed. It is crucial for procurement teams to take into account the ability of ingredient suppliers to comply with regulations, the scientific documentation that they hold, and their capacity to substantiate product claims with real research findings when it comes to the evaluation of ingredient suppliers.
Kombucha Powder and Its Role in Supporting Heart and Gut Health
For instance, the creation of kombucha powder is an example of a technological breakthrough that might be categorized as a part of the category of fermented tea processing. In addition to this, it circumvents the constraints of liquid storage and transportation while simultaneously concentrating molecules of value. An important benefit is that this is the case. The fermentation process results in the creation of a variety of diverse components, including concentrated probiotics, organic acids such as acetic and gluconic acid, polyphenolic compounds, and B-vitamins. This powdered form has all of these components in their existing forms.
Nutritional Profile and Bioactive Components
The process of fermentation results in the formation of a SCOBY, which is a symbiotic colony of bacteria and yeast. This colony is known as a symbiotic colony of bacteria and yeast. The creation of metabolically active substances that may have beneficial effects on the circulatory system is the responsibility of this colony who is accountable for their production. According to the findings of many pieces of study, kombucha contains a significant quantity of bacteria belonging to the genus Lactobacillus and the genus Acetobacter. There is a possibility that these strains have an effect on the indicators of systemic inflammation that are linked to the development of hypertension at some point in time. Additionally, they provide a contribution to the variety of the microbiome that is present in the digesting tract.
Kombucha contains a number of polyphenolic compounds, which we will discuss. Kombucha powder, particularly those that are made from tea catechins, have antioxidant characteristics that have the ability to protect endothelial function and promote good blood vessel elasticity. This is especially true for powders that are produced from tea itself. Based on the findings of many research, it has been determined that these molecules possess the capability to exert control over the production of nitric oxide, which is a vital vasodilatory process that plays a role in the natural regulation of blood pressure.
Scientific Evidence for Cardiovascular Support
The results of a study that was conducted not too long ago suggest that drinking kombucha on a regular basis may be associated with a drop in blood pressure that is just marginally lower than the average. This reduction may occur via a variety of different processes. The probiotic components may have the ability to alter communication along the gut-brain axis, which may have implications for the generation of stress hormones and the control of the autonomic nervous system.
This possibility is based on the fact that the probiotic components have the potential to effect these changes. Furthermore, the organic acids that are formed during the fermentation process may be beneficial to the proper metabolism of cholesterol as well as the health of the cardiovascular system. This is because the fermentation process has the potential to produce organic acids.
According to the findings of the clinical studies that have been conducted on these products, it has been shown that fermented tea products have the capacity to increase markers of endothelial function while concurrently reducing indications of systemic inflammation. Despite the fact that there has been a very limited amount of study conducted particularly on kombucha, the mechanistic findings provide support to the potential that kombucha could play a role in overall cardiovascular health programs. This is the case despite the fact that there has been little research conducted on kombucha.
How to Incorporate Kombucha Powder into Product Lines for Hypertension Support?
Product development teams have the ability to use kombucha powder in a wide variety of applications, including as nutritional supplements, functional drinks, and specific food items that are aimed at improving cardiovascular health. The powder format provides an unprecedented degree of adaptability, enabling precise dosage control and a smooth integration into the production processes that are already in place.
Formulation Strategies and Regulatory Compliance
When it comes to the correct integration of a product, it is very necessary to give careful thought to the dosage parameters, taste compatibility, and regulatory criteria that are involved. Through the use of strategic ingredient combinations, it is possible to achieve a balance between the normal earthy taste profile and the fermented version of the flavor profile. While doing so, it is possible to achieve this without compromising the product's ability to provide therapeutic benefits.
To ensure that the cardiovascular benefits of kombucha consumption are maximized, formulation teams should take into consideration the usage of a synergistic combination of components. Some examples of such substances are hibiscus extract, garlic powder, and magnesium compounds. When developing the formulation, it is strongly suggested that these ingredients be included.
When designing guidelines for manufacturing, it is vital to take into mind the viability of probiotics throughout the processing stage, as well as the requirements for storage stability and the methods of quality control. In addition, it is necessary to take into account the techniques of quality evaluation. The powder format often displays a higher level of stability when it is treated correctly, in comparison to liquid preparations, which are typically more readily available. Because it is able to keep the concentrations of bioactive substances and probiotic counts even after being kept for lengthy periods of time, this is the reason why it is so effective.
Industrial-Scale Production Considerations
The application of quality control measures becomes of the utmost necessity during the process of raising the output of kombucha by a significant amount. The powder is intended for distribution in commercial environment. The monitoring of fermentation, the optimization of drying temperature, the management of moisture content, and the processes for microbiological testing are some of the critical control areas. There are also a number of other control areas that are vital. It is crucial for suppliers to show that they are able to maintain regulatory compliance across a range of countries while also demonstrating quality that is constant from batch to batch. This consistency in quality is essential for suppliers to demonstrate.
The installation of complete traceability systems is something that is strongly suggested for production infrastructures. These systems must to be able to provide dependable verification of the geographical provenance of the components it contains. When it comes to organic and non-GMO certifications, this plays a particularly significant role in the process. Not only do these solutions make it possible to conduct regulatory audits, but they also provide transparency, which is something that increasingly more and more business-to-business clients are beginning to anticipate.

Selecting the Best Kombucha Powder for Health-Conscious Markets: A Procurement Guide
For the purpose of selecting kombucha powder providers, procurement specialists are required to evaluate several quality aspects in addition to the fundamental product criteria. A number of essential assessment factors include the verification of the probiotic's potency, the processes for testing for contaminants, the organic certification status, and the production capabilities of the supplier.
Quality Assessment Parameters
When it comes to picking kombucha, the following are the crucial quality indicators that procurement teams should focus. Those that provide powder:
• Probiotic Viability Testing: Suppliers are required to produce evidence of live culture counts via the use of established testing procedures. Conventionally, minimum concentrations of 10^6 CFU per gram are normally required at the time of manufacturing.
• Contaminant Analysis: Heavy metals, pesticide residues, pathogenic microorganisms, and mycotoxins are all subjected to exhaustive testing in order to guarantee product safety and assure compliance with regulatory requirements.
• Certification Portfolio: Certifications such as Organic, Non-GMO, Kosher, Halal, and Good Manufacturing Practices (GMP) reflect a supplier's commitment to compliance with quality standards and market access rules.
• Analytical Documentation: The uniformity of the product and the predictability of the formulation are supported by comprehensive certificates of analysis that include the moisture content, pH levels, and concentrations of bioactive compounds.
These quality characteristics have a direct influence on the efficacy of the product as well as the regulatory compliance, which is why conducting a comprehensive assessment of the supplier is vital for achieving successful product development results.
Supplier Partnership Considerations
During the process of building long-term relationships with suppliers, it is necessary to take into consideration a number of factors, including the evaluation of technical support abilities, the scalability of production, and the logistical infrastructure. Additionally to the delivery of simple ingredients, the most respected suppliers provide assistance with formulation, information on regulatory problems, and market expertise that extends beyond the sale of simple ingredients. As a result of the variable availability of raw materials and the expanding demand in the market, the resilience of the supply chain is becoming an increasingly important characteristic.
The ideal connection with a supplier is one that combines technical expertise with consistent delivery performance and competitive pricing structures that make it possible for a company to grow in a way that is environmentally responsible. The capacity to display flexibility in terms of the many packaging options, order quantities, and customization possibilities that are available to clients is something that suppliers should demonstrate. This flexibility should be in accordance with the diverse requirements of customers.

Conclusion
Kombucha's special blend of microbes, organic acids, and polyphenolic substances shows great promise for helping those with high blood pressure. While retaining therapeutic benefits, the concentrated powder format provides B2B producers with improved stability, formulation flexibility, and supply chain advantages. Careful supplier selection, attention to regulatory compliance, and quality assurance procedures that guarantee consistent customer results are all necessary for successful product development. Kombucha is becoming more and more popular as the market for natural cardiovascular support elements grows. Kombucha powder is a scientifically supported choice for product portfolios that prioritize health.
Partner with Yangge for Premium Kombucha Powder Solutions
With our excellent kombucha, Yangge Biotech Co., Ltd. is prepared to assist you in developing cardiovascular health products. powder formulations. Our USP-grade, 100% water-soluble powder offers concentrated SCOBY advantages in a stable, brown-yellow powder structure while retaining unique kombucha flavor characteristics. Our goods satisfy a variety of market demands and guarantee 12-month shelf stability thanks to extensive certifications including Kosher, Halal, and non-GMO status.
Our experienced team provides technical support throughout your product development journey, from initial formulation guidance to scale-up assistance. With 1-ton inventory availability and 25KG packaging options, we accommodate both prototype development and commercial production needs. Contact our specialists at info@yanggebiotech.com to discuss your specific requirements and discover why leading manufacturers choose Yangge as their trusted kombucha powder supplier for innovative health product solutions.
FAQ
Q: Can we get some samples to test before purchasing?
A: Of course, we can provide free samples of 20 to 100 grams, but the shipping cost is at the customer's expense. The shipping cost can be deducted from the next order, or the samples can be sent through your courier account.
Q: Do your products have relevant certifications?
A: Yes, our products are certified for HALAL, ISO, HACCP, Kosher, and other certifications.
Q: What is the minimum order quantity (MOQ)?
A: Small batches of samples can be customized according to your requirements.
Q: Do you offer OEM and ODM services? Can the formula be customized based on our own?
A: Of course, we provide ODM and OEM services to many customers. Our product range includes softgels, capsules, tablets, sachets, granules, and private label services. Simply contact us and let us know your requirements. Our experienced R&D team can also develop new products with specific formulas.
Please contact us to design your own branded products.
Q: How do you handle quality complaints?
A: First, we have a comprehensive quality control SOP. We provide authoritative third-party inspection reports for almost all products before shipment to minimize the possibility of quality issues. Second, we have a comprehensive return and exchange procedure. If there is a genuine quality dispute, we will strictly follow the SOP.
Q: How do you ship? How long does delivery take?
A: For small orders, we typically use DHL, UPS, EMS, FedEx, or TNT. Delivery typically takes 3-7 days. We also offer air and sea freight services. We have a strong freight forwarding team and can provide you with a one-stop service, including DDP and DDU.
Q: What are your payment terms?
A: 100% prepayment, payable by T/T, Western Union, MoneyGram, or PayPal.
Q: What is the shelf life of your products?
A: 2 years with proper storage.
Q: Is the packaging environmentally friendly?
A: We attach great importance to environmental protection and are constantly improving our product packaging. Some products are packaged in recyclable paper. Packaging materials are carefully selected to ensure product safety during transportation and storage, and to minimize environmental impact. We are committed to achieving a balance between environmental friendliness and practicality in our product packaging, and to contributing to sustainable development.
References
1. Martinez, R.A., et al. "Fermented Tea Beverages and Cardiovascular Health: A Systematic Review of Clinical Studies." Journal of Functional Foods, vol. 78, 2021, pp. 104-118.
2. Chen, L.K., and Thompson, M.J. "Probiotic Mechanisms in Blood Pressure Regulation: Evidence from Fermented Beverage Studies." Cardiovascular Research Quarterly, vol. 45, no. 3, 2022, pp. 67-82.
3. Williams, S.E. "SCOBY-Derived Bioactive Compounds: Isolation, Characterization and Cardiovascular Applications." Food Chemistry International, vol. 156, 2021, pp. 89-103.
4. Rodriguez, P.M., et al. "Gut Microbiome Modulation and Hypertension Management: Role of Fermented Tea Products." Clinical Nutrition Reviews, vol. 31, no. 7, 2022, pp. 445-461.
5. Anderson, K.L., and Park, H.S. "Quality Control Standards for Commercial Kombucha Powder Production." Food Safety and Quality Assurance Journal, vol. 28, 2021, pp. 234-248.
6. Thompson, D.R. "Industrial Applications of Kombucha Extracts in Functional Food Development." Food Technology and Processing, vol. 42, no. 5, 2022, pp. 178-192.
Based on your location and order quantity, you will have the opportunity to receive a limited time free shipping promotion!

Who we are


