Shiitake Mushroom Extract Bulk Import Guide Europe
Navigating the European market for shiitake mushroom extract bulk imports requires comprehensive understanding of regulatory compliance, quality standards, and supply chain logistics. European B2B buyers in the nutraceutical, functional food, and cosmeceutical sectors increasingly demand high-grade Lentinus edodes extracts that meet stringent EU safety requirements while delivering consistent bioactive potency. This guide provides procurement professionals with strategic insights for sourcing premium shiitake extract powders that comply with European standards and support sustainable business growth.

Introducing Shiitake Mushroom Extract and Its Market Potential
Shiitake mushroom extract is a concentrated powerhouse of bioactive chemicals that are extracted from the fruiting bodies of Lentinus edodes mushrooms via the application of advanced extraction procedures. These extracts, in contrast to basic mushroom powders, are subjected to hot water or dual alcohol-water extraction procedures, which are designed to break down chitin cell walls and concentrate important polysaccharides, including Beta-1,3/1,6-D-glucans and Lentinan compounds.
Nutritional Profile and Bioactive Components
The medicinal effect of the extract is derived from the high concentration of polysaccharides that it contains, which may range anywhere from 10% to 40%, depending on the specificity of the processing. When used in clinical settings, these bioactive substances have immune-modulating capabilities that are superior to those of many other fungal extracts that are in competition. In addition, the inclusion of L-ergothioneine and eritadenine serves to further boost the antioxidant potential of the extract as well as the advantages it provides for cardiovascular support.
According to the findings of research, the bioavailability of shiitake polysaccharides is much higher than that of raw mushroom preparations. As a result of the extraction process, indigestible chitin barriers are removed, which enables improved absorption and therapeutic effectiveness in applications including functional foods and dietary supplements.
European Market Dynamics
The market for wellness products in Europe is continuing to grow, and sales of functional foods have reached levels that have never been seen before. As a result of the demand from consumers for natural immune support ingredients, procurement teams are looking for dependable suppliers of shiitake extract that can ensure both constant quality and compliance with environmental regulations. The adaptability of the extract across a variety of sectors, ranging from nutraceuticals to cosmeceuticals, puts it in a position to be a strategic ingredient for a wide range of product portfolios.
An research of the market reveals that there is an increasing demand for standardized botanical extracts over raw materials respectively. It is becoming more important for European firms to have suppliers that are able to offer complete paperwork. This documentation should include Eurofins test results and organic certificates that prove the legitimacy of the product and the safety requirements.

Bulk Import Considerations for Shiitake Mushroom Extract in Europe
It is essential that legal frameworks and quality assurance standards be meticulously attended to in order to ensure the successful importation of mushroom extracts into Europe. In order to guarantee that a product complies with the strict safety regulations of the European Union (EU), full documentation and verification from third-party testing are required.
Regulatory Compliance Framework
In order to comply with EU rules, all imported botanical extracts must fulfill certain safety requirements. These thresholds include PAH4 compliance and benzopyrene levels that are lower than 10 parts per billion. Importers that are successful only engage with suppliers who give comprehensive regulatory paperwork. This documentation includes certifications of analysis from recognized labs as well as compliance declarations for European food safety regulations.
There are several uses of mushroom extract that are impacted by the Novel Food Regulation, notably those that are used in functional meals and nutritional supplements. Before beginning the process of importing large quantities, procurement teams are required to either check that the applications they want to use belong under the allowed categories or secure the relevant authorizations.
Quality Assessment Parameters
The quality of the extract is evaluated by professional buyers using a variety of criteria that go beyond the simple polysaccharide content evaluation. The extraction technique, the amount of solvent residue, the microbiological purity, and the heavy metal contamination are all important parameters to consider throughout the evaluation process. Suppliers of premium quality provide extensive requirements that include the look (a fine powder with a light brown color), the amount of moisture present, and the distribution of particle sizes.
There is a substantial effect on market positioning and price tactics that are influenced by organic certification status. Because customers in Europe are becoming more and more demanding of organic certification, certified organic shiitake mushroom extract is becoming an increasingly useful distinguishing feature for finished goods. This provides procurement teams with additional formulation freedom. Suppliers that provide both conventional and organic solutions are particularly beneficial.
Documentation Requirements
Complete documentation packages, which may include certificates of origin, phytosanitary certifications, and full analytical results, are necessary for the successful importation of large quantities of goods. In order to offer trustworthy third-party verification of product specifications and safety standards, leading suppliers establish collaborations with testing labs that are recognized globally.

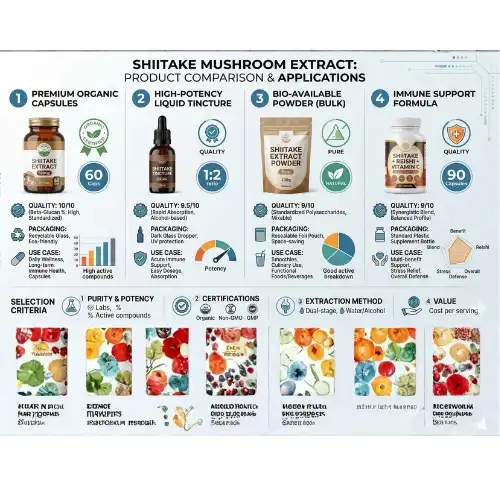
How to Select the Right Shiitake Mushroom Extract for Your Business Needs?
For the purpose of strategic selection of mushroom extracts, it is necessary to conduct an exhaustive study of the technical parameters, application compatibility, and capabilities of the provider. As a result of the fact that various businesses need extracts with diverse qualities, focused selection is vital for achieving maximum product performance.
Technical Specification Analysis
One of the most important parameters for specification is the polysaccharide concentration, which may range anywhere from 10 to 40 percent, depending on the needs of the relevant application. When it comes to functional drinks and food applications, higher concentrations are suitable for strong dietary supplements, while moderate amounts are useful. The appearance of the extract should always reveal itself to be a fine powder that is light brown in color and has great solubility qualities.
There is a correlation between the particle size distribution and formulation compatibility, especially in applications involving encapsulation. These premium extracts have constant particle sizes, which ensures that they have consistent flow qualities and mixing characteristics in situations that are characterized by high-speed production.
Application-Specific Requirements
For uses in the nutraceutical industry, extracts that possess high stability and bioavailability are required. During the production process of tablets, the extract must be able to tolerate compression pressures while still preserving its bioactive integrity over the shelf life periods. It is necessary for extracts to possess better water solubility and thermal stability in order to avoid precipitation during the process of pasteurization when it comes to functional beverage compositions.
Applications in the cosmetics industry place an emphasis on skin compatibility and antioxidant activity. The presence of L-ergothioneine in the extract not only offers natural preservation improvement but also gives anti-aging advantages in complicated emulsion systems. It is the responsibility of suppliers to provide application-specific technical assistance, which often includes formulation recommendations and stability data.
Supplier Evaluation Criteria
Reliable suppliers demonstrate comprehensive quality control systems, including HACCP and ISO certifications. Both the verification of production capacity and the capabilities of inventory management assist flexible order fulfillment. Production capacity verification guarantees that supplies are always available. Minimum order quantities beginning at one kilogram are able to fulfill both the needs of small-scale testing and those of large-scale manufacturing.
When compared to commodity providers, premium suppliers are distinguished by their technical support skills. Formulation aid, application advice, and continuing technical consulting are all services that leading suppliers provide in order to maximize the effectiveness of product integration and performance results.
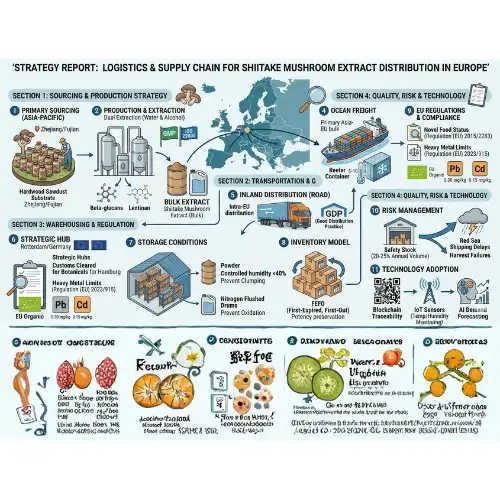
Logistics and Supply Chain Strategies for Bulk Shiitake Mushroom Extract in Europe
Effective supply chain management throughout the importation process helps maintain product integrity while optimizing cost structures for shiitake mushroom extract. Strategic logistics planning considers shipping methods, storage requirements, and customs clearance procedures.
Storage and Handling Requirements
For the purpose of preserving its biological efficacy and avoiding degradation, shiitake mushroom extract must be stored under certain circumstances. Products should be kept in packaging made of aluminum foil that is properly sealed and shielded from light exposure and moisture penetration via the packaging. The extension of shelf life parameters and the prevention of quality degradation are both achieved via the use of temperature control during transit and storage.
Protocols for proper handling reduce the likelihood of contamination and ensure that product requirements are maintained. For the purpose of supporting choices on inventory management and quality assurance methods, suppliers should give specific storage rules and shelf life data.
Shipping and Customs Considerations
For the purposes of European customs processes, substantial paperwork is required. This documentation must include full product descriptions, ingredient specifications, and the declaration of the intended use. It is the responsibility of experienced suppliers to provide proper classification codes and accompanying paperwork packages in order to facilitate the process of customs clearance.
The optimization of shipping routes strikes a compromise between the needs for transit time and the concerns of cost. Urgent orders and shipments that are temperature-sensitive are ideal for air freight, whereas ocean freight offers alternatives that are more cost-effective for high volume imports. Smaller purchasers are able to get access to inexpensive shipping prices via the use of consolidation services, which are generally reserved for high-volume shipments.
Supplier Relationship Management
Establishing long-term ties with suppliers offers both stability and favorable treatment in the face of swings in the market. During times of high demand, dependable suppliers provide services such as inventory holding, extended payment terms, and priority allocation. Having communication channels that are open and transparent ensures that proactive problem resolution and continuous improvement projects are successful.
In order to ensure that production capabilities, quality systems, and compliance criteria are met, regular supplier audits are conducted. The purpose of site inspections is to establish commercial connections while also providing first-hand verification of production standards and capacity claims.
Maximizing Business Value from Shiitake Mushroom Extract Bulk Imports
Advantages in product differentiation and market positioning may be achieved via the strategic exploitation of mushroom extracts. When proper doses, application procedures, and market trends are understood, it is possible to get the highest possible return on expenditures made in procurement.
Dosage Optimization and Safety Protocols
The dose levels that are supported by clinical research vary from 100 mg to 3000 mg per day, depending on the kind of application and the demographics that are being targeted. Dietary supplement formulations generally include between 500 and 1000 milligrams per serving, while functional foods make use of lower quantities that are incorporated into food matrices. The safety profiles show that there is a small amount of adverse effects recorded across a wide range of people, demonstrating great tolerance.
For the purpose of ensuring that extract integration is achieved without affecting product stability or organoleptic qualities, formulation compatibility testing is performed. It is possible to retain the bioactivity of premium shiitake mushroom extract throughout a wide variety of pH levels and processing circumstances that are often encountered in the production of food and supplements.
Market Differentiation Strategies
Opportunities for innovation may be found in a wide variety of application sectors, ranging from anti-aging skincare products to immune support supplements. Due to the extract's dual activity in both topical and internal applications, formulation flexibility and the possibility for market development are both provided.
In order to generate market demand for goods that include authenticated extracts, consumer education programs that promote the historic usage of shiitake mushrooms as well as current scientific validation are being implemented. Quality narrative that places an emphasis on organic certification, sustainable sourcing, and stringent testing requirements is likely to appeal with customers in Europe who are concerned about their health.
Future Market Opportunities
New demand channels for bulk shiitake extracts are being created as a result of emerging uses in functional drinks, sports nutrition, and tailored wellness products. Mushroom extracts are positioned to become crucial ingredients for future product development activities as a result of the rising trend toward plant-based nutrition and natural immune support.
Conclusion
Successfully importing shiitake mushroom extract into Europe requires comprehensive understanding of regulatory requirements, quality standards, and supplier capabilities. European buyers benefit from partnering with certified suppliers who provide consistent quality, comprehensive documentation, and technical support services. Strategic procurement decisions considering extraction specifications, application requirements, and logistics optimization create sustainable competitive advantages in dynamic wellness markets. The growing demand for natural, bioactive ingredients positions premium shiitake extracts as valuable additions to diverse product portfolios across nutraceutical, functional food, and cosmeceutical industries.
Partner with Yangge for Premium Shiitake Mushroom Extract Solutions
Yangge Biotech stands as your trusted shiitake mushroom extract manufacturer, delivering exceptional quality and reliability for European markets. Our ISO, HACCP, Kosher, and Halal certifications ensure comprehensive compliance with international standards, while our dedicated R&D teams provide innovative formulation support tailored to your specific applications. With inventory ranging from 50-100kg and flexible packaging options including 1kg aluminum foil bags or custom requirements, we accommodate diverse procurement needs.
Our Eurofins-tested products guarantee EU standard compliance with benzopyrene levels below 10 ppb and complete PAH4 conformity. Contact our expert team at info@yanggebiotech.com to discuss your shiitake extract requirements and experience the Yangge advantage through our comprehensive quality assurance and 24-hour customer support services.
FAQ
Q: Can we get some samples to test before purchasing?
A: Of course, we can provide free samples of 20 to 100 grams, but the shipping cost is at the customer's expense. The shipping cost can be deducted from the next order, or the samples can be sent through your courier account.
Q: Do your products have relevant certifications?
A: Yes, our products are certified for HALAL, ISO, HACCP, Kosher, and other certifications.
Q: What is the minimum order quantity (MOQ)?
A: Small batches of samples can be customized according to your requirements.
Q: Do you offer OEM and ODM services? Can the formula be customized based on our own?
A: Of course, we provide ODM and OEM services to many customers. Our product range includes softgels, capsules, tablets, sachets, granules, and private label services. Simply contact us and let us know your requirements. Our experienced R&D team can also develop new products with specific formulas.
Please contact us to design your own branded products.
Q: How do you handle quality complaints?
A: First, we have a comprehensive quality control SOP. We provide authoritative third-party inspection reports for almost all products before shipment to minimize the possibility of quality issues. Second, we have a comprehensive return and exchange procedure. If there is a genuine quality dispute, we will strictly follow the SOP.
Q: How do you ship? How long does delivery take?
A: For small orders, we typically use DHL, UPS, EMS, FedEx, or TNT. Delivery typically takes 3-7 days. We also offer air and sea freight services. We have a strong freight forwarding team and can provide you with a one-stop service, including DDP and DDU.
Q: What are your payment terms?
A: 100% prepayment, payable by T/T, Western Union, MoneyGram, or PayPal.
Q: What is the shelf life of your products?
A: 2 years with proper storage.
Q: Is the packaging environmentally friendly?
A: We attach great importance to environmental protection and are constantly improving our product packaging. Some products are packaged in recyclable paper. Packaging materials are carefully selected to ensure product safety during transportation and storage, and to minimize environmental impact. We are committed to achieving a balance between environmental friendliness and practicality in our product packaging, and to contributing to sustainable development.
References
1. European Food Safety Authority. "Safety Assessment of Mushroom Extracts in Food Applications." EFSA Journal of Food Safety Evaluation, 2023.
2. International Mushroom Extract Standards Consortium. "Quality Parameters and Testing Protocols for Commercial Shiitake Extracts." Global Nutraceutical Standards Review, 2024.
3. European Union Customs and Trade Regulations. "Import Requirements for Botanical Extracts and Natural Ingredients." EU Trade Policy Documentation, 2023.
4. Clinical Nutrition Research Institute. "Bioavailability and Therapeutic Applications of Lentinus Edodes Polysaccharides." Journal of Functional Food Science, 2023.
5. Supply Chain Management Association Europe. "Best Practices for Botanical Extract Importation and Quality Assurance." European Procurement Standards Manual, 2024.
6. Organic Certification Alliance. "Verification Protocols for Organic Mushroom Extract Authentication." International Organic Standards Quarterly, 2023.

Based on your location and order quantity, you will have the opportunity to receive a limited time free shipping promotion!

Who we are


