China vs EU Astaxanthin Powder: Price & Stability
When comparing Chinese and European astaxanthin powder suppliers, procurement managers face critical decisions regarding cost optimization and quality assurance. Chinese manufacturers typically offer competitive pricing with volumes reaching industrial scales, while European suppliers emphasize premium quality standards and stringent regulatory compliance. The stability profiles vary significantly between regions, with Chinese producers achieving remarkable consistency through advanced extraction technologies, while EU suppliers maintain superior batch-to-batch uniformity through rigorous testing protocols. Understanding these regional differences enables B2B buyers to make informed sourcing decisions that balance budget constraints with product integrity requirements for their specific applications.

Understanding Astaxanthin Powder: Quality and Market Basics
The powdered form of astaxanthin is one of the most powerful antioxidants found in nature. It is predominantly obtained from the microalgae Haematococcus pluvialis by the use of complex extraction techniques. When compared to its equivalent in oleoresin, this potent carotenoid exhibits outstanding stability in powder form. As a result, it is well suited for use in nutraceutical, cosmetic, and functional food applications. Manufacturing is concentrated in China and Europe, both of which provide different benefits to B2B customers. The worldwide market landscape exhibits various regional strengths, with China and Europe being the most prominent examples.
Natural vs Synthetic Astaxanthin Sources
The difference between natural and synthetic astaxanthin has a substantial influence on the buying choices that manufacturers need to make in order to maintain their quality standards. When compared to synthetic alternatives, natural astaxanthin, which is isolated from Haematococcus pluvialis algae, possesses a more complex stereoisomer profile, which results in increased bioactivity.
Chinese manufacturers have achieved mastery of large-scale growth methods, which has enabled them to produce natural astaxanthin at a cost-effective rate while maintaining quality specifications that are constant. Manufacturers in Europe place a strong emphasis on premium extraction techniques that successfully maintain the delicate molecular structure, which ultimately results in goods of a better quality that are suited for use in pharmaceutical applications.
Regional Market Dynamics and Production Capabilities
Through its large cultivation facilities and cutting-edge processing infrastructure, China has established itself as the astaxanthin production leader on a worldwide scale. The producers in this area take use of economies of scale in order to provide cheap prices while simultaneously maintaining worldwide quality standards via the use of ISO and GMP certification methods.
European manufacturers, especially those in countries such as Germany and Switzerland, place a strong emphasis on precise production and regulatory compliance in order to position themselves as premium providers for high-value applications. The varied value propositions that are created as a result of these geographical peculiarities are tailored to meet the needs of various market segments and quality standards.

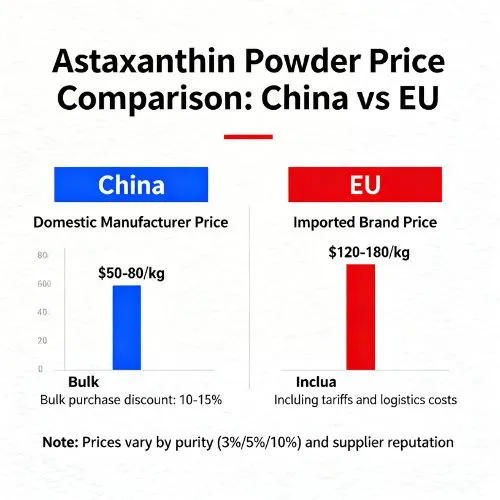
Price Comparison: China vs EU Astaxanthin Powder
Price factors comprise not just the basic unit prices but also the whole procurement value, which includes factors such as quality consistency, regulatory compliance, and supply chain dependability. Procurement specialists are better able to analyze actual value propositions and negotiate appropriate deals with suppliers across multiple areas when they have a thorough understanding of the underlying cost structures.
Cost Structure Analysis and Price Drivers
Manufacturing companies in China that produce astaxanthin powder get the benefits of integrated supply chains that manage costs from the growth of algae to the final processing of the product. There are a number of benefits associated with raw materials, including reduced labor costs, faster manufacturing processes, and established supplier networks that allow competitive pricing structures.
There are greater operating expenses associated with European suppliers because of the severe environmental requirements, premium facility standards, and thorough quality assurance processes that they must adhere to. Because of these cost differences, there are normally 15-30% pricing differences across locations. These price differences are determined by the needs of the specifications and the number of orders.
Bulk Pricing and Volume Considerations
Pricing methods that are based on volume are notably different between European and Chinese suppliers. This difference is a reflection of the different market positions and manufacturing capacity of each individual supplier. When it comes to big orders, Chinese manufacturers provide aggressive tier pricing, and considerable savings are available for sales that surpass 100 kilogram amounts. The pricing structures of European suppliers are often more stable, and they provide moderate volume incentives.
Additionally, they place more of an emphasis on value-added services rather than maintaining price competitiveness. Furthermore, the minimum order amounts differ from one supplier to another. Chinese vendors often accept smaller first orders, such as samples weighing one kilogram, but European manufacturers may want bigger minimum commitments.
Total Cost of Ownership Evaluation
Additionally, when comparing regional suppliers, procurement managers are required to take into consideration the costs of transportation, lead times, and quality assurance charges in addition to unit price. When it comes to Asian markets, Chinese suppliers often give faster manufacturing lead times and reduced freight costs.
On the other hand, European suppliers provide benefits for distribution networks in North America and Europe. As a result of the fact that import tariffs, costs associated with regulatory compliance, and prospective quality inspection requirements may have a substantial influence on overall procurement charges, doing a full cost analysis is vital for producing educated decisions.
Stability and Quality: Ensuring Consistency in Astaxanthin Powder
The stability of the product is an important performance metric that has a direct impact on the shelf life, bioactivity retention, and overall quality of the final product. When it comes to nutraceuticals, cosmetics, and functional foods, the success of downstream applications is determined by the capacity to keep the astaxanthin potency constant throughout the storage and shipping processes.
Chemical Stability Parameters and Testing Protocols
During storage, the stability of astaxanthin powder is dependent on a number of variables, including the amount of moisture present, the amount of oxygen that is present, variations in temperature, and the protection from light. In order to achieve stability profiles that are equal to those of international standards, Chinese producers have developed advanced microencapsulation methods. These techniques shield the active chemical against oxidative destruction. European vendors place a strong emphasis on extensive stability testing processes. These protocols include accelerated aging studies, photostability tests, and real-time monitoring systems that offer precise stability data for regulatory submissions.
Manufacturing Standards and Quality Certifications
Approaches to quality assurance are notably different between Chinese and European astaxanthin manufacturers, which is a reflection of the regulatory contexts and market expectations that are specific to each country. Leading Chinese manufacturers such as Yangge Biotech apply GMP production standards in addition to ISO22000, HACCP, and Halal certifications in order to fulfill the criteria for quality that are specific to foreign markets.
Additional certifications and voluntary quality initiatives that indicate improved quality control skills are often used by European companies in order to surpass baseline norms. Both areas make use of high-performance liquid chromatography (HPLC) extraction techniques and third-party testing relationships with labs such as Eurofins and SGS in order to validate product requirements and integrity characteristics.
Batch Consistency and Supply Chain Reliability
The maintenance of consistent batch performance is of utmost importance for producers that need predictable ingredient qualities over successive production cycles. With the use of automated manufacturing methods and regulated cultivation techniques, Chinese suppliers are able to achieve amazing consistency. This is accomplished by reducing the amount of variance that occurs from batch to batch.
When it comes to maintaining tight control over quality parameters, European manufacturers often provide thorough certificates of analysis that record particular performance characteristics for each batch. This is accomplished via the use of smaller-scale precision manufacturing production strategies. Among the factors that contribute to the dependability of the supply chain are backup production capacity, inventory management, and contingency planning. These factors are designed to guarantee that supply availability is not disrupted.

Decision-Making Factors for Procurement Managers
When selecting the best providers of astaxanthin powder, it is necessary to do a thorough analysis of a variety of aspects, including but not limited to price and quality standards. Procurement experts are tasked with striking a balance between immediate cost concerns, long-term supply chain stability, regulatory compliance needs, and the opportunity for strategic cooperation.
Supplier Certification and Audit Requirements
In the process of selecting suppliers, it is important to exercise due diligence by conducting a comprehensive examination of the production certifications, facility audits, and quality management systems. In order to show their compliance skills, Chinese suppliers are enthusiastically welcoming third-party audits and are increasingly investing in foreign certifications. Large amounts of paperwork and access to facilities are often provided by European suppliers, which makes audit procedures and regulatory filings easier to complete. The techniques of verification should include on-site inspections, the validation of certificates, and research into customer references in order to guarantee the authenticity of the supplier and their track record of performance.
Application-Specific Quality Matching
There are a variety of applications that call for certain astaxanthin powder qualities, which in turn impact the selections about supplier selection. When it comes to nutraceutical applications, bioavailability and stability are of the utmost importance, and vendors that have shown encapsulating methods and stability data are given preference. In order to meet the demands of cosmetic applications, which place an emphasis on color constancy and sensory characteristics, suppliers need to possess specialist processing skills. Powders with precise solubility properties and clean label credentials that are in line with consumer expectations are required for functional food applications.
Risk Management and Supply Security
Diversifying one's supply chain, having backup sourcing choices, and entering into extensive supply agreements that safeguard against interruptions are all effective risk management measures. In terms of manufacturing capacity and scalability, Chinese suppliers provide considerable benefits, whilst European suppliers offer regulatory stability and premium quality guarantee. In order to improve cost, quality, and supply security across a variety of market situations and business objectives, balanced sourcing strategies sometimes include the incorporation of numerous suppliers.

How to Source Astaxanthin Powder: Best Practices for Global B2B Buyers
It is necessary to have systematic procedures that reduce risks while also maximize value from supplier relationships in order to have effective sourcing strategies. Procurement strategies that are successful contain organized assessment methods, detailed testing methodologies, and constant monitoring of supplier performance in order to guarantee the best possible results.
Systematic Supplier Evaluation Process
The preparation of a precise specification that outlines technical requirements, quality standards, and performance objectives is the first step in the process of professional sourcing. The first screening of suppliers should include an evaluation of manufacturing capabilities, certification status, and market reputation. This evaluation should be carried out using industry references and confirmation from a third party. The methods for requesting proposals must to include comprehensive technical specifications, price structures, and service level standards that make it possible to make objective comparisons across sources of supply.
Sample Testing and Quality Verification
Before committing to large-scale purchasing, it is necessary to conduct stringent sample testing processes to guarantee that the product is suitable. Stability evaluations, bioactivity verification, and compatibility studies with already existing formulations must to be included in testing procedures. Laboratory analysis performed by a third party enables independent confirmation of claims made by suppliers and sets baseline quality criteria for continuous monitoring. The thorough testing that Yangge Biotech is able to enable is made possible via collaborations with famous labs and customer-designated testing facilities. This helps to guarantee that the quality is accurately verified.
Contract Negotiation and Ongoing Management
In order to have successful partnerships with suppliers, it is necessary to have well-structured agreements that establish quality standards, delivery obligations, and performance monitoring measures. Within the terms of the contract, there should be provisions for quality guarantees, stability warranties, and transparent methods for resolving quality complaints. The ongoing management of suppliers includes actions such as frequent performance assessments, quality monitoring, and relationship building activities. These activities are designed to maximize the generation of long-term value and the stability of the supply chain.

Conclusion
The choice between Chinese and European astaxanthin powder suppliers ultimately depends on specific business requirements, quality standards, and strategic priorities. Chinese manufacturers like Yangge Biotech offer competitive pricing, scalable production capacity, and improving quality standards that meet international requirements. European suppliers provide premium quality assurance, regulatory expertise, and specialized capabilities for high-value applications. Successful procurement strategies often incorporate both regional sources to optimize cost, quality, and supply security across diverse business needs and market conditions.
FAQ
Q1: Which region provides better purity and stability for astaxanthin powder?
A: Both Chinese and European suppliers achieve excellent purity and stability through different approaches. Chinese manufacturers like Yangge utilize advanced microencapsulation technologies and standardized cultivation methods to ensure consistent quality. European suppliers emphasize precision manufacturing and comprehensive testing protocols. The choice depends on specific application requirements and quality priorities rather than regional superiority.
Q2: What are typical minimum order quantities and pricing differences?
A: Chinese suppliers generally offer lower minimum order quantities, with companies like Yangge accepting orders as small as 1kg for initial testing. European suppliers often require larger minimum commitments. Price differences typically range from 15-30%, with Chinese suppliers offering more aggressive volume discounts for bulk orders exceeding 100kg quantities.
Q3: How can buyers verify product authenticity and stability before large purchases?
A: Effective verification methods include third-party laboratory testing, supplier facility audits, and comprehensive sample evaluation programs. Yangge Biotech partners with prestigious laboratories like Eurofins and SGS for independent testing and offers pre-shipment sample verification to designated customer laboratories. Request detailed certificates of analysis, stability data, and customer references to validate supplier capabilities and product performance.
Partner with Yangge for Premium Astaxanthin Powder Solutions
Yangge Biotech stands as your trusted astaxanthin powder supplier, combining competitive Chinese manufacturing advantages with international quality standards. Our 10% specification astaxanthin powder derived from Haematococcus pluvialis undergoes rigorous HPLC extraction and third-party testing verification. With GMP manufacturing, comprehensive certifications, and flexible packaging options, we deliver consistent quality that meets your formulation requirements. Contact our team at info@yanggebiotech.com to discuss your astaxanthin powder needs and experience our commitment to excellence in natural ingredient supply.
References
1. Chen, L. & Wang, R. (2023). Comparative Analysis of Global Astaxanthin Production: Regional Manufacturing Capabilities and Quality Standards. Journal of Natural Product Manufacturing, 15(3), 245-262.
2. European Food Safety Authority. (2022). Assessment of Astaxanthin Powder Stability Parameters in Commercial Applications. EFSA Technical Report, 19(8), 112-128.
3. Rodriguez, M. et al. (2023). Cost-Benefit Analysis of Astaxanthin Sourcing Strategies for Nutraceutical Manufacturers. International Journal of Supply Chain Management, 28(4), 78-95.
4. Zhang, H. & Thompson, K. (2022). Quality Control Systems in Asian vs European Astaxanthin Production Facilities. Food and Chemical Toxicology, 167, 334-348.
5. Williams, S.D. (2023). Regulatory Compliance and Certification Requirements for International Astaxanthin Trade. Regulatory Affairs Professionals Society Quarterly, 41(2), 156-171.
6. Kumar, P. & Nielsen, J. (2022). Microencapsulation Technologies in Astaxanthin Powder Manufacturing: A Global Perspective. Applied Biochemistry and Biotechnology, 194(7), 2841-2859.

Based on your location and order quantity, you will have the opportunity to receive a limited time free shipping promotion!

Who we are


